 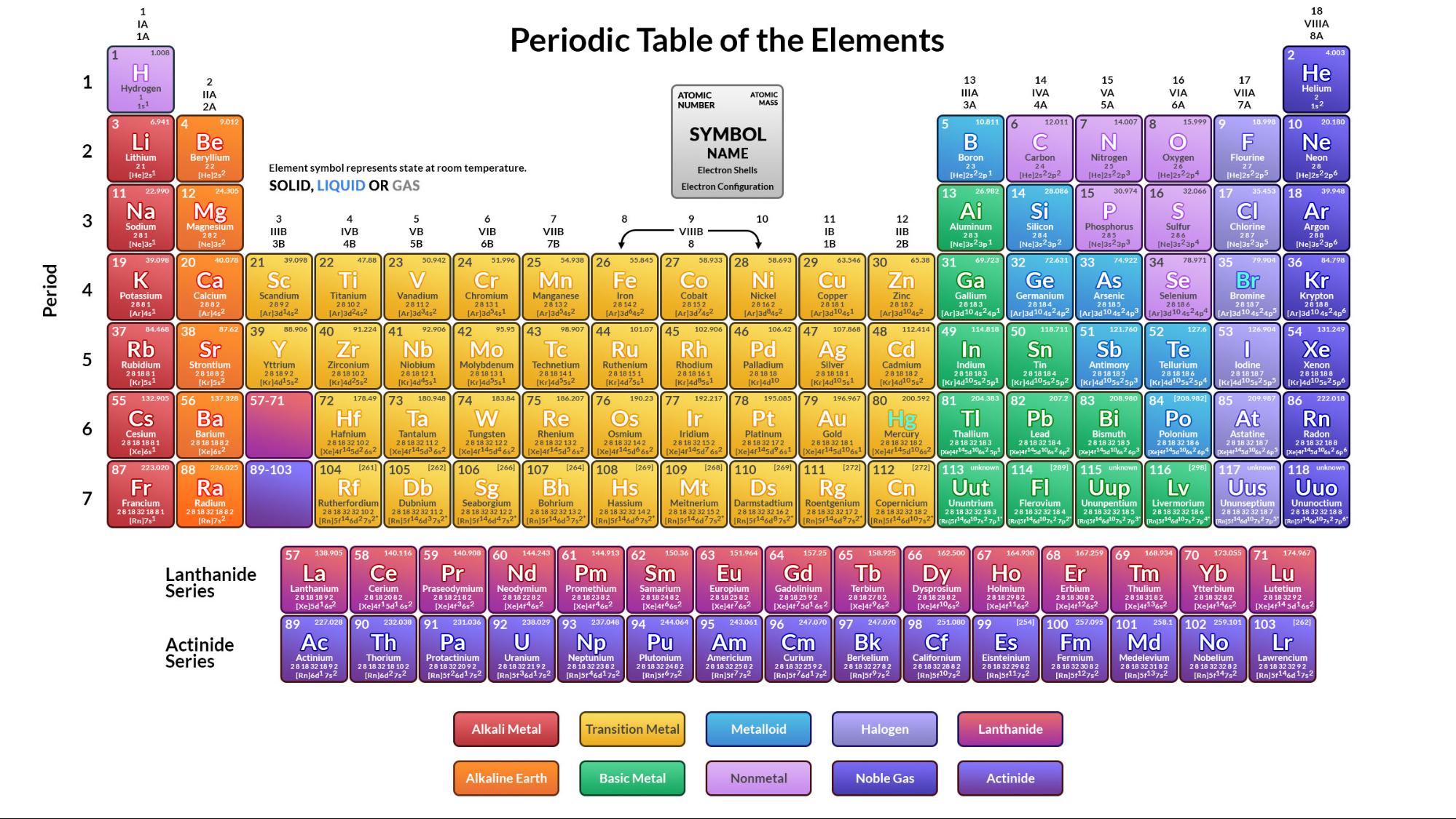
In chemistry this is done according to physical and chemical properties. 
to classify close classify To arrange something into classes according to characteristics. For example, melting point, electrical conductivity, appearance at room temperature. It is possible to use information about an element’s physical properties close physical property A property of an element or compound which can be directly observed or measured. This number is determined by the fact that in any sample of carbon there are some atoms of. Metals are found on the left and in the middle, whereas non-metals are all on the right. When looking at carbon on the periodic table, it says carbon has an average atomic mass of 12.011 amu. The periodic table can be used to find out if an element is a metal or a non-metal. Elements are listed on the periodic table. account for around 20% of known elements close element A pure substance which is made from only one type of atom. Non-metal elements are on the right hand side of the periodic table. are metals, while non-metals close non-metal A substance that has the typical properties of a non-metal. It allows us to spot patterns and make predictions about other elements. 
Most elements in the periodic table close periodic table A table which lists all of the chemical elements and arranges them in a way that is useful. Gethin: They would just melt and it would just make a mess anyway. Gethin: So, it's great for thermometers, but not so good for pots and pans. So, when it gets hot the space between the particles expands and then this rises up the thermometer. Miss Armit: Yeah, because mercury's a great conductor of heat. Gethin: Well, it's something to do with the heat. Miss Armit: It's the only liquid transistor metal and because of its properties it's perfect for a thermometer. And gold and silver are used in jewellery, because they're shiny, they're malleable and unreactive with oxygen, so they don't rust easily. We've also got iron and aluminium and they're great at conducting heat, so we use these metals for our pots and pans. Miss Armit: Well, we've got copper and it's a really good conductor of electricity, so we use that in our wires. And what metals are we talking about here and what are they used for. Miss Armit: So their key properties are they conduct electricity, they conduct heat, they are malleable - which means they're bendy - and they're shiny. So we're going to talk about transition metals, which are found in the middle of the periodic table. Gethin: I think this is going to be good, isn't it. Miss Armit: So what we're going to look at today are the metal elements in Group 1 of the periodic table, along with the metal elements in the transition metal block of the periodic table and their properties. And today, we're going to be looking at the periodic table. What she doesn't know about chemistry is not worth knowing. Unfortunately, there was a slightly different system in place in Europe.Gethin: Today, we've got super science teacher Miss Armit with us. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. 
These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
